Physiology
Creatinine is produced as the result of normal muscle metabolism. Phosphocreatine, an energy-storing molecule in muscle, undergoes spontaneous cyclization to form creatine and inorganic phosphorous. Creatine then decomposes to creatinine. Note that creatine is actually produced in the liver from amino acids (glycine and arginine). In health, production and excretion of creatinine are fairly constant in an individual animal, resulting in low variation in an individual animal (Ruaux et al 2012, Hokamp and Nabity review 2016). An additional and relatively minor source is creatinine ingested during consumption of muscle tissue and absorbed from the intestines. Creatinine is filtered freely through the glomerulus and is not reabsorbed in the tubules (except in goats). Therefore, creatinine is considered a more reliable measure of GFR, compared to urea nitrogen, in most species, except for ferrets and potentially goats, as it is not influenced by diet or protein catabolism. Measurement of creatinine concentration in serum or plasma is included in chemistry profiles mainly to screen for decreased glomerular filtration rate (GFR), even though the relationship between creatinine and GFR is not linear and creatinine is relatively insensitive to early changes in GFR.
Methodology
Technique used at Cornell
Two point endpoint with creatininase and sarcosine oxidase (enzymatic method)
Procedure
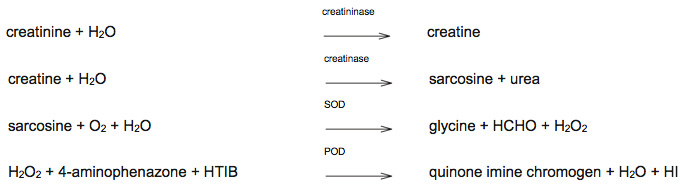
Creatininase converts creatinine to creatine and then sarcosine and urea. Sarcosine reacts with sarcosine oxidase (SOD) to form glycine, formaldehyde (HCHO) and hydrogen peroxide (H2O2). The hydrogen peroxide is converted by horseradish peroxidase (POD) in the presence of a dye (p-aminophenazone or PAP) and 2,4,6-triiodo-3-hydroxybenzoic acid (HTIB) to a colored chromogen (quinone), which is directly proportional to the creatinine concentration, and hydrogen iodide (HI) (image is copied from product information sheet).
There are chromogen-based reactions for creatinine measurement, where creatinine reacts with picric acid. This is the so-called Jaffe reaction. We used to use the modified or kinetic version of the Jaffe reaction to measure creatinine in our laboratory but have moved to the enzymatic (PAP) method above.
Units of measurement
Creatinine concentration is measured in mg/dL (conventional units) and μmol/L (SI unit). The conversion formula is shown below:
mg/dL x 88.4 = μmol/L
Sample considerations
Sample type
Serum, heparin or EDTA plasma, and urine
Anticoagulant
Lithium heparin or K2-EDTA
Stability
The stability of creatinine in human serum and plasma is as follows: 7 days at 15 – 25°C or 2 – 8°C, and 3 months frozen (-15 to -25°C) (Roche product information sheet)
Urine should be collected in the absence of any additives. If preservatives for other analytes are required, use only hydrochloric acid or boric acid. The stability of creatinine levels in urine samples are as follows (per Roche product information sheet):
- Stability in urine (without preservative): 2 days at 15 – 25°C, 6 days at 2 – 8°C, and 6 months frozen (-15 to -25°C).
- Stability in urine (with preservative): 3 days at 15 – 25°C, 8 days at 2 – 8°C, and 3 weeks frozen (-15 to -25°C).
Interferences
- Enzymatic method: These were obtained off the Roche product information sheet.
- Lipemia (intralipid): No substantial interference up to 2000 lipemic index.
- Hemolysis: May increase with severe hemolysis (>800 hemolysis index)
- Icterus: Increases in unconjugated bilirubin >20 mg/dL or conjugated bilirubin >15 mg/dL or an icteric index >15 units may falsely decrease creatinine concentrations. This interference has not been clearly documented in animals, although studies of dogs with Babesia canis (de Scally et al 2004) and icteric cases seen here have had unexpectedly low creatinine concentrations.
- Jaffe method
- Hemolysis, icterus, lipemia: May interfere with results similar to the enzymatic but at lower index values than that indicated above for the enzymatic method (based on our previously used assay, e.g. < 4 mg/dL for bilirubin).
- Binding of non-creatinine chromogens: Non-creatinine chromogens that react with picric acid are reported to be glucose, vitamin C, proteins, acetone (>50 mg/dL) acetoacetate (> 20 mmol/L), β-hydroxybutyrate (> 25 mmol/L), pyruvate, and cephalosporin-based drugs. These are expected to falsely increase values. In a spiking study of creatinine measurement in dog, cat, horse and cattle sera using a kinetic Jaffe and enzymatic method, increased values were seen with spiking with glucose, acetone, cefazolin, and and cefoxitin in all species and decreased values were seen with spiking with acetoacetic acid (possibly due to pH changes) in the kinetic Jaffe but not the enzymatic method. Interferences such as bilirubin decreased results with both assays. Ceftiofur had more of a negative effect on creatinine with the Jaffe reaction (Jacobs et al 1991). The latter study showed variable changes with hemolysis (spiked hemoglobin) and lipemia (spiked intralipid), with results tending to decrease with high amounts of lipid with both reactions. One study with human sera did not show substantial interference with β-hydroxybutyrate in a kinetic Jaffe (Kemperman et al 2000) and the degree of interference is analyzer dependent (Bowers and Wong 1980). Glucose and vitamin C can also decrease creatinine with the Jaffe reaction as they act as reducing substances (Gerard and Khayam-Bashi 1984). Vitamin C and protein do not affect the enzymatic reaction.
Test interpretation
Creatinine shows less variability within than between individuals with the enzymatic method, indicating that subject versus population-based reference intervals are preferable for interpretation (Kopke et al 2018). For example, International Renal Interest Society (IRIS) recommendations are that small changes in creatinine (>0.2 mg/dL in dogs and cats) in 48 hours in animals indicates acute kidney injury (IRIS acute kidney injury guidelines).
Increased creatinine concentration
- Artifact: When measured by the Jaffe technique, both creatinine and non-creatinine chromogens react with the reagent. Non-creatinine chromogens include acetone, pyruvate, and drugs, particularly cephalosporin-based antibiotics. When present in high concentrations, these can falsely increase creatinine values.
- Physiologic causes:
- Physiologic: Creatinine is higher in premature and newborn foals (up to 8 mg/dL in newborn foals; this is thought to be due to defective placental transfer with the allantois containing more creatinine than plasma and should decline to with in the adult reference interval within 3-5 days (Bozorgmanesh et al 2021)) and heavily muscled horses (up to 2.5 mg/dL). Data is less consistent in Holstein calves. In one study, the upper limit of a reference interval for calves of 1-14 days of age was slightly higher (1.6 mg/dL) than adult animals at or older than 2 years of age (1.4 mg/dL) (Lumsden et al 1980). In another study, mean creatinine concentrations were within reference intervals in neonatal calves (24-48 hours and up to 14 days) (Mohri et al 2007). Creatinine concentrations are higher in greyhounds (Dunlop et al 2011, Liffman et al 2018), with a published upper reference limit of 2.0 mg/dL; Dunlop et al 2011), presumably due to increased muscle mass.
- Increased production: A mild increase in creatinine (< 1 mg/dL) may be seen after ingestion of a recent cooked meat meal. A study in 100 clinically health dogs did not find any consistent increases in creatinine for up to 8 hours after ingestion of the dogs’ regular meal (Yi et al 2022).
- Pathophysiologic causes
- Decreased GFR: Creatinine will increase with azotemia or decreased GFR that is due to prerenal, renal or post-renal causes. In ruminants and horses, creatinine is a better measure of GFR than urea nitrogen (due to gastrointestinal excretion and degradation of urea). Creatinine is a fairly insensitive marker of GFR, being increased with approximately a 1/3 reduction in renal function as measured by GFR (based on inulin clearance) and a 75% decrease in renal mass (Hokamp and Nabity review 2016). Less severe reductions in GFR may not manifest with a high creatinine. Because of the high index of individuality, monitoring changes in individual animals over time may be a more sensitive indicator of declining renal function (GFR) than comparing results to a previously established reference interval, which may be too broad to detect changes in individual animals unless there are substantial reductions in GFR. A change in creatinine of more than 0.3 mg/dL is considered support of declining GFR, but this change may be within the analytical variation of many machines used to measure creatinine (Hokamp and Nabity review 2016). Hence, as for all clinical pathologic values, results should always be interpreted in context of what is known or suspected to be occurring in the patient at hand.
- Release from muscle: Although there have been reports of high creatinine due to release of creatine from muscle, studies have shown that acute myositis does not consistently increase creatinine concentrations. It is more likely that the creatinine is increased due to acute kidney injury and renal azotemia from myoglobinuric nephrosis as a consequence of myoglobin release from severe myositis or myopathy.
Decreased creatinine concentration
- Physiologic causes
- Decreased muscle mass: Creatinine trends lower in small breeds of dogs, based on body weight (Misbach et al 2014, Hokamp and Nabity review 2016). Young dogs have lower creatinine than adult dogs, presumably due to lower muscle mass (Rosset et al 2012, Rørtveit et al 2015).
- Increased GFR: This occurs during pregnancy (due to increased cardiac output).
- Pathophysiologic causes
- Decreased production: Loss of muscle mass from starvation or cachexia may decrease creatinine concentrations. In rats, there is evidence that sepsis reduces creatinine production, although the method of determination of creatinine production was indirect (Doi et al 2009). The mechanism is unclear but could be related to sepsis-associated hypothermia. Although creatine is produced in the liver, low creatinine with liver insufficiency (e.g. portosystemic shunting) is likely due to increased glomerular filtration rate (Deppe et al 1999) versus decreased creatine production in the liver.
- Increased glomerular filtration rate: This occurs in animals with portosystemic shunts. Urea nitrogen is also frequently low from decreased liver synthesis of urea in such patients.
Discordant urea and creatinine
Urea nitrogen and creatinine should always be interpreted together and in relation to the glomerular filtration rate. Below is a summary table of interpretations of different urea and creatinine combinations.
| Urea nitrogen | Creatinine | Interpretation |
| ↑ | N / ↓ | Early prerenal or renal azotemia Renal azotemia in ferrets Normal glomerular filtration rate (GFR) with non-renal causes of ↑ urea nitrogen High protein diet, upper gastrointestinal (GI) bleed, protein catabolism ↓ GFR with non-renal causes of ↓ creatinine Decreased muscle mass (cachexia), sepsis, marked hyperbilirubinemia (typically > 15 mg/dL may result in a false decrease in creatinine) |
| N / ↓ | ↑ | ↓ GFR with non-renal causes of ↓ urea nitrogen Hepatic synthetic failure, polyuria-polydipsia (in the absence of chronic kidney disease), low protein diet, metabolism of urea nitrogen by GI flora (horses and cattle) Normal GFR with non-renal causes of ↑ creatinine A normal finding in Greyhounds and other heavy muscled animals (e.g. Draft horses) (increased muscle mass), post-prandial in ferrets |
